Industry Prospective
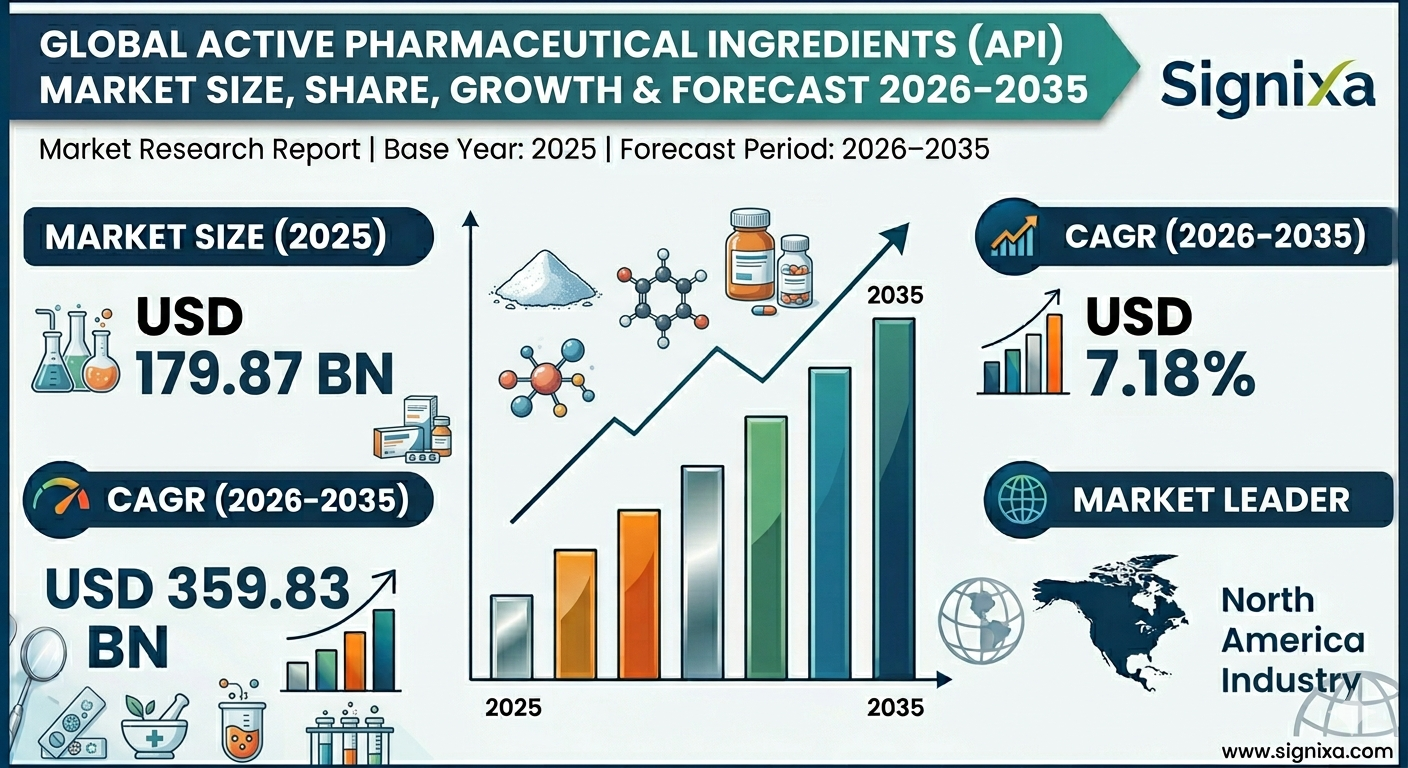
The size of the global Active Pharmaceutical Ingredients (API) market was worth around USD179.87 billion in 2025 and is projected to grow to around USD 359.83 billion by 2035, with a compound annual growth rate (CAGR) of roughly 7.18% between 2026 and 2035. The report examines key growth drivers, restraints, opportunities, and challenges across global and regional markets.
API Market Overview
The APIs market forms the core of the supply chain in producing pharmaceutical finished products. APIs offer the actual therapeutic effect of the drug. The demand for APIs is increasing because of increasing incidence of chronic diseases leading to hospitalisation, increased generics produced, blockbuster drug patent expiries, and an increased geriatric population. Compliance standards continue to become stricter. Other trends, such as outsourcing and biotechnology, also have an impact on the supply of APIs.
This expansion is primarily driven by the escalating global burden of chronic and infectious diseases, such as cardiovascular disorders, cancer, and diabetes, which necessitate continuous demand for a wide range of medications. The aging global population, with its increased susceptibility to various ailments, further contributes to the rising consumption of pharmaceutical products and, consequently, APIs.
Moreover, significant advancements in pharmaceutical research and development (R&D), coupled with increased funding from both public and private sectors, are propelling the discovery and development of new drugs, each requiring novel APIs. The growing focus on personalized medicine and targeted therapies, which often rely on highly specific and potent APIs, is also creating new opportunities for market players. While synthetic APIs currently dominate the market due to their cost-effectiveness and widespread use in generic drugs, the biotech API segment (including monoclonal antibodies, vaccines, and recombinant proteins) is projected to record the fastest growth, driven by innovations in biopharmaceuticals and their efficacy in treating complex diseases.
Despite its critical role in global healthcare, the API market faces several complex challenges. A significant hurdle is the stringent regulatory landscape governing API manufacturing. Regulatory bodies like the FDA, EMA, and WHO impose rigorous guidelines for quality control, safety, efficacy, and Good Manufacturing Practices (GMPs). Adhering to these strict standards requires substantial investment in advanced manufacturing facilities, robust quality management systems, and extensive documentation, which can be particularly challenging for smaller manufacturers. Non-compliance can lead to severe penalties, market recalls, and significant reputational damage.
Furthermore, the global API supply chain is often fragmented and complex, with a significant concentration of manufacturing in regions like China and India due to lower production costs. This over-reliance on a few geographical hubs creates vulnerabilities, as demonstrated by supply chain disruptions during the COVID-19 pandemic, leading to shortages and price volatility for essential APIs. Geopolitical tensions, trade disputes, and environmental regulations in key manufacturing countries can also impact supply stability. The intense competition within the generic API segment puts continuous pressure on pricing, forcing manufacturers to balance cost-efficiency with uncompromising quality, while the intellectual property landscape for innovative APIs remains a complex challenge for both branded and generic drug producers.
Key Findings
- The global API market is expected to grow at a CAGR of 7.18% from 2026 to 2035.
- The market size is forecasted to increase from USD179.87 billion in 2025 to USD 359.83 billion by 2035.
- The innovative API segment holds a significant market share due to increased R&D spending and novel drug approvals.
- The generic API segment is witnessing robust growth fueled by global healthcare cost-containment policies.
- Merchant API manufacturers dominate the market as outsourcing trends rise.
- Synthetic APIs currently lead the market, though biotech APIs are gaining rapid traction.
- Oncology is the leading application segment, followed by cardiovascular and CNS disorders.
- North America leads the market, with Asia-Pacific emerging as the fastest-growing region.
API Market Dynamics
Rising Chronic Disease Burden, Aging Population & Generic Drug Demand
The increasing worldwide prevalence of chronic and lifestyle diseases is a major driver of API market expansion. Aging populations and unhealthy lifestyles contribute to higher demand for medications for diabetes, cardiovascular diseases, and cancer, leading to an increased volume of active pharmaceutical ingredients, especially in therapeutic categories with growing drug consumption trends.
Another key driver is the increasing demand for generic medicines. The expiry of patents on blockbuster drugs is boosting the production of generic APIs. All regional and local governments are urging the use of generics to try and cut healthcare costs. Thus, the demand for generic APIs is escalating rapidly, particularly in developing countries such as India and Brazil, where affordable healthcare is essential to the population.
The API market's dynamics are fundamentally shaped by the ever-increasing global demand for effective and affordable medications, stemming from factors like the rising incidence of chronic and infectious diseases and an aging global population. This demographic shift significantly boosts the consumption of various therapeutic drugs, directly driving the need for their active components. Furthermore, the relentless pace of pharmaceutical R&D is a powerful dynamic, as new diseases emerge and scientists uncover novel therapeutic pathways, driving demand for innovative APIs including highly specialized APIs for personalized medicine and targeted therapies.
Biotech API Revolution -Monoclonal Antibodies, Biologics & Personalized Medicine
Technology advances in biotech-diverse API synthesis continue to revolutionize the space. Monoclonal antibodies, recombinant proteins, and cellular-based therapies are growing in utilization, and the use of biotech APIs is on the rise. The increasing complexity of these drugs requires advanced manufacturing technologies, which in turn is driving investments in biopharmaceutical production capacities.
Beyond medical necessity, economic drivers play a crucial role. The expiry of patents for blockbuster drugs is a significant dynamic, opening vast opportunities for generic drug manufacturers. As these patents lapse, the demand for high-volume, cost-effective generic APIs surges, leading to intense competition among API producers. The expansion of healthcare access and rising disposable incomes in emerging markets also create substantial opportunities for API manufacturers, as these regions represent untapped growth potential for pharmaceutical consumption. This interplay of global health needs, scientific innovation, and economic accessibility continuously reshapes the demand landscape for APIs.
GMP Compliance, Regulatory Stringency & FDA/EMA Quality Standards
The API market also navigates a complex set of dynamic challenges, particularly concerning regulatory stringency and supply chain vulnerabilities. The highly regulated nature of API manufacturing, with stringent Good Manufacturing Practices (GMPs) and quality control standards imposed by global authorities like the FDA and EMA, is a constant dynamic. While crucial for patient safety, these regulations necessitate significant investments in compliant facilities, advanced quality systems, and extensive documentation, which can deter new entrants and increase operational costs for existing players. Non-compliance carries severe penalties, including market bans and reputational damage, making regulatory adherence a paramount concern.
API Supply Chain Vulnerabilities, Reshoring & Geopolitical Risks
Furthermore, the concentration of API manufacturing in a few key geographical regions, predominantly China and India, represents a critical supply chain dynamic. While these regions offer cost advantages, this over-reliance has exposed the market to significant vulnerabilities, as demonstrated during the COVID-19 pandemic. Disruptions due to natural disasters, geopolitical tensions, or health crises in these manufacturing hubs can lead to widespread shortages and price volatility for essential medicines globally. This has spurred a dynamic of reshoring or diversifying API production closer to end-markets in North America and Europe, driven by national security and supply chain resilience concerns. Balancing cost-efficiency with geopolitical risks and ensuring supply chain robustness are ongoing challenges that significantly influence investment decisions and market strategies within the API sector.
API Market Segmentation Analysis
By Type Segment Analysis: Innovative APIs vs. Generic APIs
Innovative APIs
Innovative APIs had a considerable share of more than 55% in 2025, which is mainly due to very high-value drug innovations using extensive R&D. Innovative APIs are used for patented drugs and in the treatment of complex diseases such as autoimmune disorders and cancer. These APIs typically command premium pricing, reflect the extensive R&D investment required for novel drug discovery, and are the primary products of major global pharmaceutical companies.
Generic APIs
Generic APIs will grow more than 8.2% CAGR during the forecast period, primarily due to significant demand for cheaper alternatives and loss of exclusivity of blockbuster drugs in therapeutics such as oncology and cardiovascular medicine. Government policies promoting generic substitution to reduce healthcare costs are a major tailwind globally, particularly in high-volume markets like India, Brazil, and Southeast Asia.
By Manufacturer Segment Analysis: Merchant vs. Captive Manufacturers
Merchant Manufacturers
Merchant API manufacturers will dominate the market with nearly 65% share of global API supply in 2025. The trend of API outsourcing to third parties in countries like India and China supports this trend. These manufacturers serve multiple pharmaceutical clients and produce APIs for commercial sale, benefiting from economies of scale, specialized manufacturing expertise, and competitive pricing that drives outsourcing decisions globally.
Captive Manufacturers
Captive manufacturers still play an important role, but are increasingly targeting niche, high-potency APIs (HPAPIs) or biologics that require tight quality control and regulatory compliance. These in-house production operations allow pharmaceutical companies to maintain proprietary control over their most critical or sensitive active ingredients, particularly for complex biologic therapies and controlled substances that demand maximum quality oversight.
By Synthesis Segment Analysis: Synthetic APIs vs. Biotech APIs
Synthetic APIs
Synthetic APIs accounted for about 74% of total market revenue in 2025. Synthetic APIs remain cost-effective and broadly applicable to a wide array of therapeutic areas, specifically on the side of generics. They are produced through chemical synthesis processes and form the backbone of the global generic pharmaceutical industry, serving high-volume demand across cardiovascular, CNS, endocrinology, and gastrointestinal therapeutic areas.
Biotech APIs
The biotech API space is expanding rapidly and estimated to grow at a CAGR of 9.1% between 2025 and 2035. The rising tide of monoclonal antibodies, personalized therapies, and biologicals has been increasing demand for biotechnical-based API use worldwide. These APIs include recombinant proteins, vaccines, cell and gene therapy components, and antibody-drug conjugates — representing the frontier of modern pharmaceutical innovation.
By Application Segment Analysis: Therapeutic Areas
Oncology
Largest application segment, driven by the global rise in cancer incidences and demand for targeted therapies and immunotherapies.
Cardiovascular Diseases
Second-largest segment due to the global burden of heart conditions, hypertension, and diabetes-related cardiovascular complications.
CNS & Neurology
Growing rapidly as aging populations increase awareness of Alzheimer's, Parkinson's, and other prominent neurological diseases.
Orthopaedic
Rising demand from aging populations experiencing musculoskeletal disorders and joint-related conditions.
Endocrinology
Emerging segment with healthy CAGR rates, driven by the global diabetes epidemic and thyroid disorder prevalence.
Pulmonology
Growing demand from COPD, asthma, and post-COVID respiratory condition management across all age groups.
Gastroenterology
Inflammatory bowel disease and GI disorder prevalence driving steady API demand in this category.
Nephrology
Chronic kidney disease linked to diabetes and hypertension is fueling consistent demand for renal APIs.
Ophthalmology
Age-related macular degeneration and diabetic retinopathy are expanding the ophthalmic API market.
 Others
Dermatology, infectious diseases, immunology, and rare disease orphan drugs represent further growth segments.
Report Attributes & Market Scope
| Report Attribute | Details |
|---|---|
| Market Size Value in 2025 | USD179.87 Billion |
| Market Size Value in 2035 | USD 359.83 Billion |
| CAGR (2026–2035) | 7.18% |
| Base Year Used for Estimation | 2025 |
| Historic Data | 2020-2025 |
| Forecast Period | 2026-2035 |
| Segments Covered -By Type |
|
| Segments Covered -By Manufacturer |
|
| Segments Covered - By Synthesis |
|
| Segments Covered -By Application | Cardiovascular Diseases, Oncology (26% share), CNS & Neurology, Orthopaedic, Endocrinology, Pulmonology, Gastroenterology, Nephrology, Ophthalmology, Others |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Major Countries Covered | U.S., Canada, Mexico, Germany, UK, France, Italy, Spain, Russia, China, Japan, South Korea, India, Southeast Asia Countries, Brazil, Argentina, GCC Countries, Turkey, Iran, Israel, South Africa, Egypt, Nigeria, etc. |
| Key Companies Profiled | Teva Pharmaceutical Industries Ltd., Pfizer Inc., Novartis International AG, Dr. Reddy's Laboratories, Aurobindo Pharma, Sun Pharmaceutical Industries, Lonza Group, BASF SE, Cambrex Corporation, and Zhejiang Medicine Co. Ltd. |
API Market Regional Analysis
North America continues to hold the top spot in the API market, given its advanced healthcare infrastructure, high spending in R&D, and large pharmaceutical companies. The US continues to dominate, producing API products leading the race in innovative API products and Biotech. Europe remains strong, focusing on regulatory compliance and safety. The Asia-Pacific region (India and China), has shown the largest growth due to cost-effective manufacturing, generics manufacturing in abundance and growing pharmaceutical exports. Latin America, and the Middle East & Africa, are growing markets, and also showing significant promise for development, as the healthcare system allows access, and interest in investment for domestic drug manufacturers.
North America (Market Leader)
North America holds a substantial share of the global API market, driven by its robust healthcare infrastructure, significant R&D investments, and high demand for advanced and specialty pharmaceuticals. The United States, in particular, is a major consumer and innovator in the API space. The increasing prevalence of chronic diseases like diabetes, cardiovascular disorders, and cancer, coupled with an aging population, fuels a consistent demand for a wide range of medications. There's a strong focus on innovative APIs for novel drug formulations and precision medicine, with considerable funding directed towards developing therapies for specific genetic mutations and rare disorders.
The North American API market is also witnessing a trend towards "reshoring" or diversifying API manufacturing closer to home, driven by lessons learned from supply chain vulnerabilities exposed during recent global events. This aims to enhance supply chain resilience and reduce reliance on overseas production. While synthetic APIs currently dominate the market due to the high demand for generic drugs, the biotech API segment is experiencing rapid growth, supported by significant investments in biopharmaceuticals and the development of complex biologics. Stringent regulatory frameworks from agencies like the FDA also ensure high quality and safety standards, contributing to a premium market segment for compliant APIs.
Asia-Pacific (Fastest Growing)
The Asia Pacific region is the largest and fastest-growing market for Active Pharmaceutical Ingredients globally, playing a dual role as both a major production hub and a rapidly expanding consumer market. Countries like China and India dominate API manufacturing due to cost-effective production, abundant raw materials, and skilled labor. This has historically made them primary suppliers of generic APIs to the rest of the world. The growth in demand within the region itself is fueled by massive populations, increasing healthcare expenditure, rising prevalence of chronic and infectious diseases, and expanding access to healthcare services.
The booming generic drug industry in countries like India, driven by patent expirations of branded drugs, creates a consistent and high volume demand for synthetic APIs. Simultaneously, there's a growing investment in biotechnology and pharmaceutical R&D across the region, leading to increasing demand for innovative and biotech APIs. Governments in Asia Pacific are actively promoting domestic pharmaceutical manufacturing and R&D through various initiatives and incentives, aiming to achieve greater self-sufficiency in API production and reduce reliance on imports for essential medicines. This dynamic interplay of cost-efficient production, escalating domestic demand, and strategic government support positions Asia Pacific as the undeniable powerhouse of the global API market.
Europe
Europe represents a mature yet dynamic API market, characterized by its well-established pharmaceutical industry, advanced research capabilities, and stringent regulatory environment. The region's demand for APIs is primarily driven by the increasing prevalence of chronic diseases, an aging population, and a strong focus on patient safety and quality control. European pharmaceutical companies, particularly in Germany, France, and the UK, are significant players in both innovative and generic API manufacturing. Germany, for instance, has a long-standing reputation for producing high-quality active pharmaceutical ingredients and maintains a leading market share in Europe.
While Europe historically relied heavily on API imports, particularly from Asia, there's a growing strategic imperative to increase domestic API production and reduce supply chain dependencies, especially post-pandemic. This is evident in increased investments in local manufacturing capacities. The European market also shows a rising demand for biotech APIs, driven by the expanding biopharmaceutical industry and the development of complex biologics for conditions like cancer and autoimmune disorders. Compliance with the strict regulations of the European Medicines Agency (EMA) is paramount, ensuring that APIs meet high standards of quality, safety, and efficacy, thus shaping production methods and market access.
Middle East & Africa
The Middle East & Africa (MEA) API market is an emerging region with significant growth potential, albeit from a smaller base compared to other continents. The growth is primarily driven by increasing healthcare expenditure, improving healthcare infrastructure, and a rising awareness of public health in many countries across the region. In the Middle East, particularly the Gulf Cooperation Council (GCC) countries, governments are heavily investing in diversifying their economies, including boosting the pharmaceutical sector to reduce reliance on oil and gas. This involves establishing local pharmaceutical manufacturing facilities and, consequently, increasing the demand for APIs.
In Africa, while challenges such as limited healthcare budgets and underdeveloped infrastructure persist in some areas, the rising prevalence of chronic diseases and infectious diseases, along with a growing population, is gradually driving the need for more medicines and their active ingredients. Many countries in the MEA region heavily rely on API imports, mainly from Asia, making them vulnerable to global supply chain disruptions. However, there's a growing impetus for local API manufacturing capabilities to enhance pharmaceutical security and affordability. Investments in pharmaceutical R&D and strategic partnerships aimed at technology transfer are key trends that are expected to shape the future growth and self-sufficiency of the API market in the Middle East and Africa.
Latin America
Latin America is a growing market for APIs, showing significant promise for development as the healthcare system allows access, and interest increases in investment for domestic drug manufacturers. Brazil and Mexico are the largest API consumer markets in the region, supported by sizable generic pharmaceutical industries and increasing healthcare expenditure. Government initiatives promoting local pharmaceutical manufacturing and reducing dependence on imports are creating new opportunities for API producers. Rising prevalence of chronic diseases and expanding public healthcare coverage are further driving API demand across the region.
Recent Developments in the API Market
-
2023
Pfizer announced an expansion of its API manufacturing facilities in Ireland to support innovative therapies, reinforcing its commitment to in-house production of complex APIs for its biopharmaceutical pipeline and bolstering supply chain resilience for its key oncology and vaccine products in the European and North American markets.
-
2022
Dr. Reddy's Laboratories launched several new generic APIs for oncology and CNS therapeutic areas, expanding its generics portfolio to capitalize on patent expiry opportunities in high-growth therapeutic segments and strengthen its position as a leading global merchant API supplier.
-
2021
Novartis signed a strategic agreement with a biotech CDMO to boost biologics API production, signaling a broader industry trend toward strategic outsourcing of complex biologic manufacturing to specialized contract development and manufacturing organizations (CDMOs) as demand for monoclonal antibodies and other biologics continues to accelerate.
API Market: Competitive Landscape
The global API market is rife with competition, with important players including companies such as Teva Pharmaceutical Industries Ltd., Pfizer Inc., Novartis International AG, Dr. Reddy's Laboratories, Aurobindo Pharma, Sun Pharmaceutical Industries, Lonza Group, BASF SE, Cambrex Corporation, and Zhejiang Medicine Co. Ltd. Companies are putting a lot of investment into a number of strategies to make their production facilities better, including strategic collaborations, vertical integration, and expanding to geographical areas that provide a potential for better production activity. Leading players in the API market are finding potential avenues for growth through a focus on biosimilars, high-potency APIs, and sustainable technologies for manufacturing. Companies are utilizing the ever-changing and evolving marketplace to create a level of differentiation to capture a larger market share.
Key Companies Profiled
Global API Market Segmentation Summary
By Type
By Manufacturer
By Synthesis
By Application
By Region
Frequently Asked Questions (FAQs): Active Pharmaceutical Ingredients Market
What is an Active Pharmaceutical Ingredient (API)?
An Active Pharmaceutical Ingredient (API) is the biologically active component in a pharmaceutical drug that produces the intended therapeutic effect. APIs are used in medications for various conditions, including cardiovascular diseases, cancer, and neurological disorders.
Which key factors will influence the API market growth over 2026-2035?
The global API market is expected to grow due to the increasing prevalence of chronic diseases, rising demand for generic drugs, advancements in biotechnology, and the expansion of outsourcing to merchant manufacturers for cost efficiency.
What will be the value of the API market during 2026-2035?
According to the study, the global API market was valued at approximately USD179.87 billion in 2025 and is projected to reach around USD 359.83 billion by 2035.
What will be the CAGR value of the API market during 2026-2035?
The CAGR value of the global API market is expected to be around 7.18% during the forecast period from 2026 to 2035.
Which region will contribute notably towards the API market value?
The global API market is led by North America, supported by strong R&D infrastructure and pharmaceutical innovation. Asia-Pacific is emerging as the fastest-growing region due to expanding manufacturing capacity, cost advantages, and favorable government policies.
Which are the major players leveraging the API market growth?
Key players in the global API market include Teva Pharmaceutical Industries Ltd., Pfizer Inc., Novartis AG, Dr. Reddy's Laboratories, Aurobindo Pharma, Sun Pharmaceutical Industries, Merck KGaA, BASF SE, Cipla Inc., and Lupin Limited.
What can be expected from the global API market report?
The report explores key aspects of the API market, including an in-depth analysis of current trends, market drivers, and restraints. It also provides a detailed segmentation by type, manufacturer, synthesis method, application, and region, along with forecasts, competitive landscape insights, and strategic developments.

